Organic Chemistry: Structure, Series, and Reactions 🧪
1. Introduction & Core Definitions
Organic Chemistry: Study of carbon compounds, excluding simple carbon oxides (CO, CO₂), carbonates (Na₂CO₃), and cyanides (KCN).
Organic Compound: Any compound containing carbon, usually bonded to hydrogen and often to O, N, or halogens.
Hydrocarbon: Organic compounds containing only carbon and hydrogen atoms.
Functional group: Atom or group of atoms responsible for the chemical properties of a compound, e.g., -OH, -COOH, -NH₂.
2. The Homologous Series
A homologous series is a family of organic compounds with the same functional group and general formula. Successive members differ by a CH₂ unit.
Physical properties (boiling/melting points) change gradually with increasing molecular mass, while chemical properties remain similar.

Diagram illustrating homologous series: alkanes, alkenes, and alkynes.
| Series | Functional Group | General Formula | Nomenclature Suffix |
|---|---|---|---|
| Alkanes | C–C single bond | CnH2n+2 | -ane |
| Alkenes | C=C double bond | CnH2n | -ene |
| Alkynes | C≡C triple bond | CnH2n-2 | -yne |
| Alcohols | -OH (Hydroxyl group) | CnH2n+1OH | -ol |
| Carboxylic Acids | -COOH | CnH2n+1COOH | -oic acid |
3. Alkanes: Saturated Hydrocarbons
Alkanes contain only single bonds (saturated) and are relatively unreactive. They are major components of petroleum and natural gas.
Alkyl group (formed when one H is removed): CnH2n+1, e.g., CH4 → CH3 (methyl)
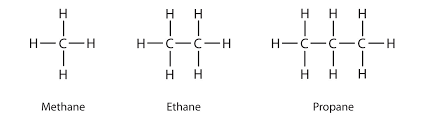
Example of an alkane structure (Methane and Ethane).
Reactions of Alkanes
A. Combustion 🔥
Complete combustion:
CH4 + 2O2 → CO2 + 2H2O
2C2H6 + 7O2 → 4CO2 + 6H2O
Incomplete combustion produces CO or soot: C3H8 + 3O2 → 3C + 4H2O

B. Halogenation (Substitution)
Reaction with halogens under UV light:
CH4 + Cl2 → CH3Cl + HCl
C2H6 + Br2 → C2H5Br + HBr

4. Alkenes: Unsaturated Hydrocarbons
- General formula: CnH2n
- Simplest member: Ethene, C2H4
- Reactions: Addition, polymerization, combustion
A. Addition Reactions
Reaction with bromine water (test for unsaturation):
C2H4 + Br2 → C2H4Br2 (1,2-dibromoethane)

Hydration (acid-catalyzed) to form alcohols:
C2H4 + H2O → CH3CH2OH
B. Polymerization
n C2H4 → [-CH2-CH2-]n (polyethene)

5. Alcohols
Alcohols have a hydroxyl (-OH) group attached to a carbon chain. General formula: CnH2n+1OH

Reactions
Combustion: C2H5OH + 3O2 → 2CO2 + 3H2O
Oxidation: CH3CH2OH + [O] → CH3CHO + H2O → CH3COOH

6. Industrial Processes
A. Cracking
Breaking long-chain hydrocarbons into smaller ones:
Catalytic cracking: C10H22 → C6H14 + C4H8 (Al2O3 catalyst)
Thermal cracking: C12H26 → C8H18 + C4H8 (high temperature)

B. Saponification
Alkaline hydrolysis of fats/oils:
Fat (triglyceride) + NaOH → Glycerol + Soap (sodium salt of fatty acid)
C3H5(COOCH2CH3)3 + 3NaOH → C3H5(OH)3 + 3CH3CH2COONa
