Sulphur and Its Compounds 🧪
1. Introduction
Sulphur is a non-metal element found in period III, group VI of the periodic table. Its electronic configuration is 2:8:6.
Sulphur occurs naturally as underground deposits and is extracted mainly using the Frasch process.
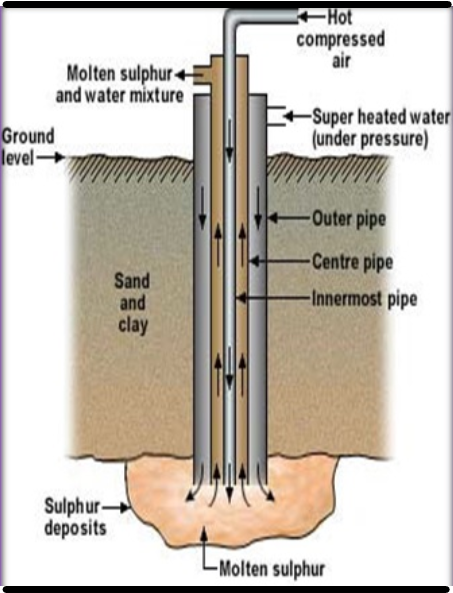
2. Frasch Process
- A hole ~30 cm in diameter is drilled through the soil to reach the sulphur bed.
- A sulphur pump made of three concentric steel pipes is lowered into the hole.
- Superheated water (~170°C, 10 atm) is forced through the outer pipe to melt sulphur.
- Hot compressed air (~15 atm) is blown through the innermost pipe to force molten sulphur to the surface.
- The sulphur is prevented from solidifying by the temperature of superheated water.
- The molten sulphur is pumped to the surface, allowed to cool, and solidified in large tanks.
- Sulphur is separated from the sulphur-water emulsion by decantation.
- The sulphur obtained is ~99% pure.
3. Important Sulphur Compounds
- Hydrogen Sulphide (H₂S): Colourless, poisonous gas with rotten egg smell.
- Sulphur Dioxide (SO₂): Colourless gas used as bleach and preservative.
- Sulphur Trioxide (SO₃): Reacts with water to form H₂SO₄.
- Sulphuric Acid (H₂SO₄): Strong acid, very important in industry.
4. Sulphuric Acid (H₂SO₄)
H₂SO₄ is a strong mineral acid used in fertilizers, batteries, detergents, and chemical manufacturing.
A. Laboratory Preparation of H₂SO₄
Concentrated H₂SO₄ can be prepared in the laboratory by heating:
- K₂SO₄ + concentrated H₂SO₄ → H₂SO₄ (concentrated)
- Or Na₂SO₄ + concentrated H₂SO₄ → NaHSO₄ + H₂O
B. Industrial Manufacture of H₂SO₄
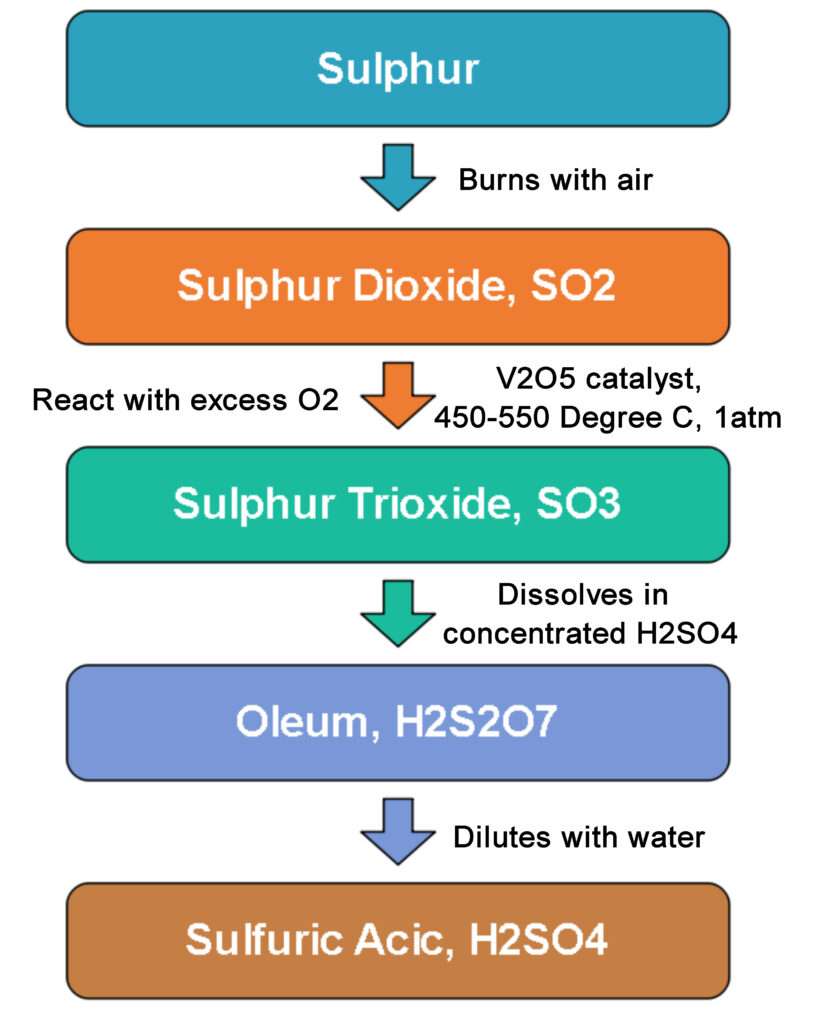
H₂SO₄ is manufactured industrially by the Contact Process in 4 main stages:
- Preparation of SO₂
- Purification of SO₂
- Catalytic oxidation of SO₂ to SO₃
- Conversion of SO₃ to H₂SO₄
The raw materials are sulphur (from Frasch process) and oxygen (from fractional distillation of liquid air).
Process:
- Burn sulphur in oxygen to produce SO₂: S + O₂ → SO₂
- Mix SO₂ with excess air and purify through dust precipitator to remove catalyst-poisoning dust and impurities (arsenic compounds).
- Dry the mixture with concentrated H₂SO₄ and pass over finely divided V₂O₅ at 450–500°C and 1 atm to form SO₃: 2SO₂ + O₂ → 2SO₃
- Dissolve SO₃ in concentrated H₂SO₄ to form oleum: SO₃ + H₂SO₄ → H₂S₂O₇
- Dilute oleum with calculated amount of water to obtain 98% pure H₂SO₄: H₂S₂O₇ + H₂O → 2H₂SO₄
NOTE I: SO₃ is first dissolved in oleum because direct dissolution in water is highly exothermic.
NOTE II: Platinum is the best catalyst but not used due to high cost and susceptibility to poisoning.
5. Physical and Chemical Properties of H₂SO₄
- Colourless, oily liquid; highly corrosive.
- Reacts with metals: 2H₂SO₄ + Zn → ZnSO₄ + H₂
- Reacts with bases: H₂SO₄ + 2NaOH → Na₂SO₄ + 2H₂O
- Reacts with carbonates: H₂SO₄ + NaHCO₃ → Na₂SO₄ + H₂O + CO₂
- Strong dehydrating agent and oxidizing agent.
6. Uses of Sulphuric Acid
- Manufacture of fertilizers (superphosphate, ammonium sulfate)
- Production of detergents and chemicals
- Lead-acid batteries
- Laboratory reagent
- Pickling of metals and industrial cleaning
7. Summary Table
| Compound | Chemical Formula | Properties | Uses |
|---|---|---|---|
| Hydrogen Sulphide | H₂S | Colourless, toxic, rotten egg smell | Laboratory reagent, metal extraction |
| Sulphur Dioxide | SO₂ | Colourless gas, bleach | Bleaching, food preservative |
| Sulphur Trioxide | SO₃ | Colourless solid, reacts with water to form H₂SO₄ | Intermediate in H₂SO₄ production |
| Sulphuric Acid | H₂SO₄ | Strong acid, dehydrating, oxidizing | Industry, batteries, fertilizers, lab reagent |